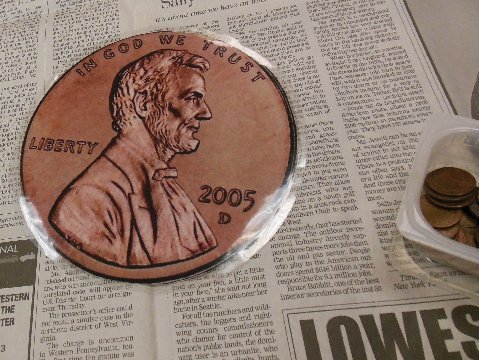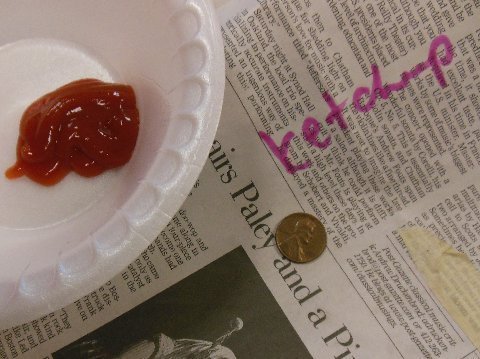Hi –
This week in Science, in honor of Presidents’ Day, we experimented with Abraham Lincoln’s head (on pennies, of course)!
First, we talked a bit about President’s Day. Who is the President? Who was the first President? Which President’s birthdays will we be celebrating next week?
Then we looked at a really big penny. Who is on it? How much money is it worth?
Did you know that Abraham Lincoln is on both sides of a penny?
Look very closely at the Lincoln Memorial on the back of a penny.
Right in the center of the building, between the 6th and 7th columns, is a statue of a seated Abraham Lincoln:
We all wanted to take a look at that, so we sat down with some magnifying glasses (which we practiced using) and some pennies.
It was difficult to see the Lincoln statue on the pennies, mostly because the pennies were quite dirty. We started guessing how to clean them and we brainstormed about ways we could make them shiny again. We had many reasonable suggestions including water, soap, milk (but we didn’t have any immediately available to try), sponges and paper towels.
We poured different liquids into separate bowls and put the bowls on the table.
Then, we each picked the liquid we thought would work the best and put our penny down next to it.
Two of us thought that water would clean the pennies.
We had one vote for soap,
two for vinegar,
five votes for soap and water,
and I was the only one who voted for ketchup. (????)
We put the pennies in the bowls and then we waited.
Finally, we took the pennies out of the bowls and wiped them off with paper towels.
Here are our results:
water
soap
vinegar
soap and water
ketchup
Wow…all of the pennies looked a little bit cleaner than before, but the ketchup really worked the best!
Weread the ingredients on the ketchup container: tomato concentrate, distilled vinegar, corn syrup, salt, spice, onion powder, natural flavors.
Hmmm…the pennies that were in the vinegar looked pretty good – let’s do one more experiment.
We stirred some salt (another ketchup ingredient) into a bowl of vinegar and then chose a ‘new’ old penny.
We held the penny so that part of it was in the solution and counted for ten seconds. Then we pulled it out.
As you can see, the right upper section came out clean!
Why does this work? The penny isn’t brown because of dirt – it is brown because over time it oxidizes and becomes covered in copper oxide. The vinegar and salt chemically react with the copper oxide and loosen it so that it can be rubbed off easily, exposing the underlying metal.